
Figure 1. Flagellated bacteria, a sketch, © E. Litvincheva
Bacteria are one of the most numerous species on Earth and constitute about 15% of the total biomass [1]. They affect the life of macroorganisms such as humans both in positive and harmful ways helping to digest the food we eat and fight pathogens, but also causing various diseases. For example, infertility, food poisoning, pneumonia, as well as many other maladies are often caused by bacterial activity. A human body of 70 kg contains about 1013 bacteria of various types weighing approximately 0.2 kg [2] (for reference, the number of human cells is estimated as also about 1013). While bacteria live throughout the human body, their main habitat is body surfaces such as skin and covered in mucus organs, e.g. gastrointestinal and reproductive tracts. To prevent bacterial infections and facilitate the proliferation of symbiotic species, we need to understand bacterial behavior. In particular, by studying bacterial motion we may find ways to control them to our advantage.
In our recently published paper [3] we aim to predict new and explain experimentally observed features of individual and collective behavior of bacteria swimming in mucus. For example, widespread bacteria Bacillus subtilis and Escherichia coli show up to 20% increase in velocity while moving back and forth following the same track [4] as if they burrow tunnels in cervical mucus thus facilitating their backward movement, see Fig. 2.

While the motion of active agents in Newtonian fluids, e.g. water, is rather well understood, complex fluids such as mucus pose substantial challenges since there are no standard well-studied mathematical models describing them. Moreover, many biofluids are anisotropic due to the presence of long biopolymer molecules like mucin in mucus. The local average orientation of these molecules (so-called “director”) significantly affects the mechanical properties of the fluid. This dependence of mucus properties on molecules orientation is a typical liquid crystalline behavior [5] as surprisingly it may sound. Thus, the mucus can be described using approaches similar to the modeling of liquid crystals utilized in common LCDs. Moreover, it is equally important that mucus shows viscoelastic properties when the relation between strain rate and stress changes in time, therefore demonstrating memory effects.
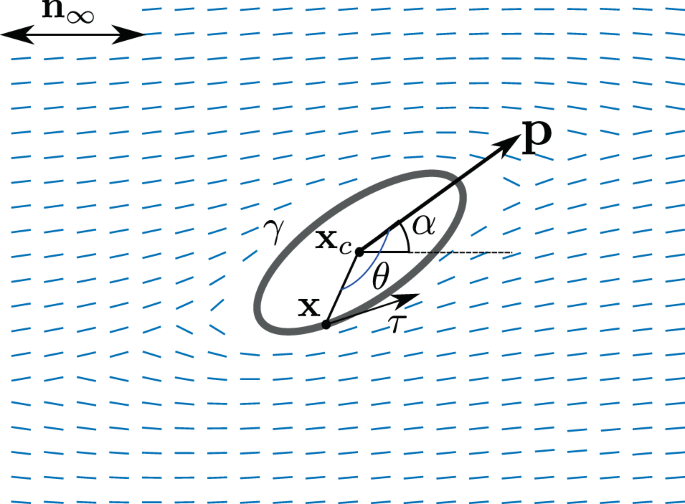
In our study [3], we utilized a model of viscoelastic liquid crystals [6] that involves nonlinear partial differential equations describing the orientation of molecules (the director) and anisotropy level (what fraction of molecules has this orientation), as well as viscoelastic stresses and fluid velocity. The coupling between liquid crystalline and viscoelastic properties is controlled by a parameter χ that, as it turned out, may have a significant influence on bacterial motion. We expanded the model [6] by adding finite-size bacteria. The real bacteria are covered in pili, tiny hair-like organelles, and some of them have flagella, similar to long tails/arms, see Fig. 1, which work as propellers or long arms stroking the fluid. We consider a simplified “microswimmer” model where a bacterium is taken as a rigid two-dimensional elongated ellipse while the activity of flagella is represented via a special (“squirmer”) boundary condition on this elliptical surface in the fluid (Stokes) equation.

For the physical parameters agreeing to the physical experiment, we observed computationally that indeed, a microswimmer in such a medium swims backward faster than forward following the same track, see Fig. 3.

In our results, a microswimmer created a zone in its wake, where anisotropy is suppressed (that is mucus behaves there like a viscous or, more precisely, viscoelastic fluid), see Fig 4. Creating the wake corresponds to burrowing tunnels in mucus in the physical experiments. When the microswimmer swims back, it travels through this zone with higher velocity than during the forth motion. This speeding up is controlled by the coupling parameter χ in an interesting way, which cannot be observed for usual viscoelastic fluids without liquid crystalline properties. Keeping all other viscoelastic parameters fixed but making |χ| larger, we prevent the director from returning to its initial state for some time. That is, we prevent the fluid in the tunnel from returning into its ordered liquid crystalline state. This is why it is crucial for our model to have coupled viscoelastic and liquid crystalline properties. It is natural to expect then, that if two microswimmers travel one after another, the follower will swim with a higher speed than the leader. The leader “burrows” the tunnel facilitating the swimming of the follower. This is the same as what we observed in another series of computational experiments. Moreover, we found that there exists a critical distance between microswimmers which again depends on the coupling parameter—the stronger the viscoelasticity, the closer bacteria may approach each other.
However, an even more intriguing behavior is obtained when two microswimmers do not follow the same trajectory. In this case, we tried to model the formation of bacterial trains. Similarly to ants following the same trail, the bacteria with intersecting trajectories may change directions of their motion so that they swim one after another. This type of behavior was observed in [7] where bacteria formed such structures in lyotropic (water-based) biocompatible liquid crystals, see Fig. 5. Such a phenomenon is specifically important since we can observe the onset of collective motion when two independent agents begin to “work” together as a team. By studying collective behavior rather than an individual one, we may learn how to control groups of bacteria.

In our study [3], the microswimmers starting positions are such that one of them crosses the wake of another. We observed that the swimmer in the wake makes a turn, wobbles around the trajectory of the leader, and then follows it in a steady manner, see Fig. 6. The same coupling parameter as before controls the spatial period of wobbling as the more viscoelastic is the mucus, the stiffer and harder to penetrate the walls of the tunnel burrowed by the leader are. So, the follower just bounces off the walls.

Based on these and other results in [3], we may predict that viscoelasticity in bacterial liquid crystalline suspensions facilitates the formation of bacterial trains and improves their stability, thus, paving the way to the onset of collective motion while allowing for bacteria to be closer to each other.
Our next goal is to modify our computational model to study specifically the collective motion of tens and hundreds of bacteria swimming in mucus. Since in our current model we exactly implement the squirmer boundary conditions on the microswimmer surface, we must introduce a fine mesh on it. That leads to prohibitively large computations if several swimmers are considered. We plan to replace rigid particles with dipoles modeling the flow that bacteria create stroking the fluid. The usage of dipoles significantly simplifies computations, and the modeling of the bacterial motion will take approximately the same time as the modeling of the mucus without bacteria.
References
[1] Bar-On YM, Phillips R, Milo R (2018) The biomass distribution on Earth, PNAS 115:6506–6511.
[2] Sender R, Fuchs S, Milo R (2016) Revised estimates for the number of human and bacteria cells in the body, PLOS Biology 14(8): e1002533.
[3] Chi H, Gavrikov A, Berlyand L, Aranson I (2022) Interaction of microswimmers in viscoelastic liquid crystals, Commun Phys 5, 274.
[4] Figueroa-Morales N, Dominguez-Rubio L, Ott TL, Aranson I (2019) Mechanical shear controls bacterial penetration in mucus, Sci Rep 9, 9713.
[5] Davies JM, Viney C (1998) Water–mucin phases: conditions for mucus liquid crystallinity, Thermochim Acta 315(1): 39–49.
[6] Hemingway EJ, Maitra A, Banerjee S, Marchetti MC, Ramaswamy S, Fielding SM, Cates ME (2015) Active viscoelastic matter: From bacterial drag reduction to turbulent solids, Phys Rev Lett 114:098302.
[7] Zhou S, Tovkach O, Golovaty D, Sokolov A, Aranson IS, Lavrentovich OD (2017) Dynamic states of swimming bacteria in a nematic liquid crystal cell with homeotropic alignment, New J Phys 19(5):055006.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in